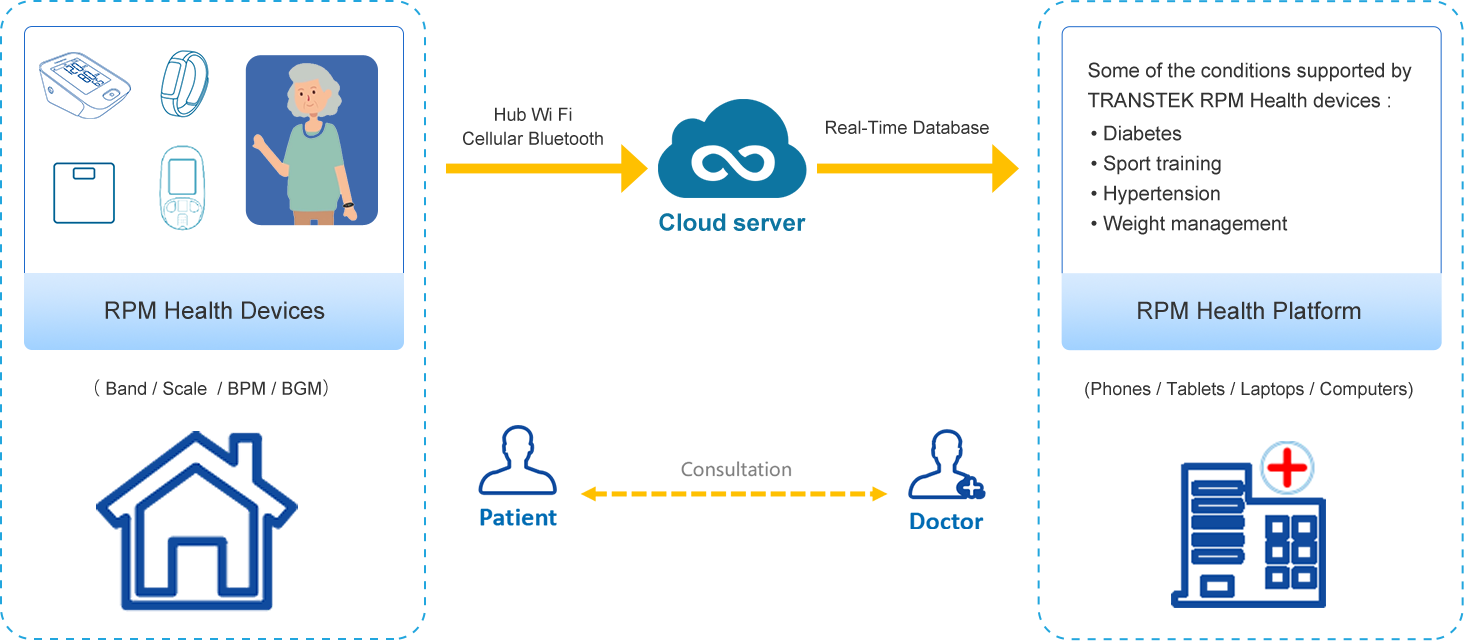
Transtek – Reliable and Market-leading RPM Devices Provider
The popularity of telemedicine has been on the rise since the outbreak of the pandemic, it brings in new models of care along remote patient monitoring (RPM) of key health metrics to improve chronic care management. As an early remote monitoring company, Guangdong Transtek Medical Electronics is on a mission to assist care providers to improve chronic care management including hypertension and diabetes by combining technological expertise and credibility. Nearly 20 years of experience in the remote monitoring healthcare industry, Transtek is responsible for meeting the growing demands of RPM and provides remote patient management, remote health monitoring, remote healthcare services, remote patient monitoring solutions and remote patient care for patients in need.